With respect to molecular diffusion, dispersion occurs as a result of an unequal concentration of the introduced material throughout the bulk medium. This process is facilitated by molecular diffusion and convection. Also, the sudden appearance of conductivity in a system of a dispersed conductive phase in an insulating matrix has been explained.ĭispersion is a process by which (in the case of solid dispersing in a liquid) agglomerated particles are separated from each other, and a new interface between the inner surface of the liquid dispersion medium and the surface of the dispersed particles is generated. This is the reason why some liquid dispersions turn to become gels or even solid at a concentration of a dispersed phase above a critical concentration (which is dependent on particle size and interfacial tension). Both surfaces have to be created (which requires a huge amount of energy), and the interfacial tension (difference of surface tension) is not compensating the energy input, if at all.Įxperimental evidence suggests dispersions have a structure very much different from any kind of statistical distribution (which would be characteristics for a system in thermodynamic equilibrium), but in contrast display structures similar to self-organisation, which can be described by non-equilibrium thermodynamics. To understand the formation and properties of such dispersions (incl emulsions), it must be considered that the dispersed phase exhibits a "surface", which is covered ("wet") by a different "surface" that, hence, are forming an interface (chemistry). In the following, only such dispersions with a dispersed phase diameter of less than 1 µm will be discussed. There are only very few studies about the structure of dispersions (emulsions), although they are plentiful in type and in use all over the world in innumerable applications (see below). However, percolation theory can be applied only if the system it should describe is in or close to thermodynamic equilibrium. Therefore, for dispersions, usually percolation theory is assumed to appropriately describe their properties. In general, dispersions of particles sufficiently large for sedimentation are called suspensions, while those of smaller particles are called colloids and solutions.ĭispersions do not display any structure i.e., the particles (or in case of emulsions: droplets) dispersed in the liquid or solid matrix (the "dispersion medium") are assumed to be statistically distributed. The two phases may be in the same or different states of matter.ĭispersions are classified in a number of different ways, including how large the particles are in relation to the particles of the continuous phase, whether or not precipitation occurs, and the presence of Brownian motion. Ī dispersion is a system in which distributed particles of one material are dispersed in a continuous phase of another material. Note 1: Modification of definition in ref. 
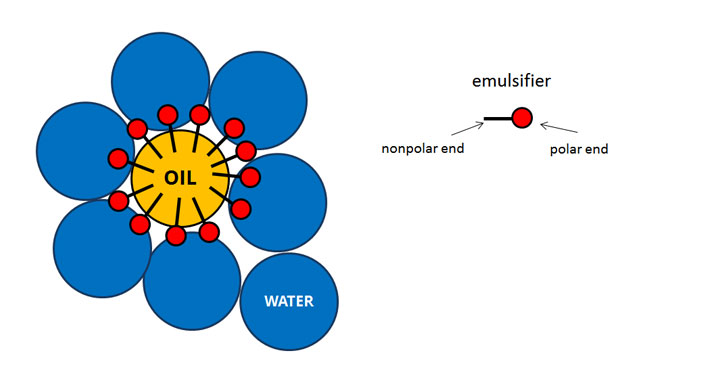
Get your taste buds ready for some fun!ĭon't forget to try out the activities featured in our STEAM Activity Chart to extend your knowledge about emulsions.Material comprising more than one phase where at least one of the phases consists of finely divided phase domains, often in the colloidal size range, dispersed throughout a continuous phase. Let's explore both temporary and permanent emulsions and how they can enhance simple Asian recipes. Dressings that use eggs or dairy products as a base, such as ranch, Caesar or blue cheese, are permanent emulsions that do not need shaking before use. Simple dressings that are made from oil, water and seasonings are temporary emulsions, which is why they must be given a good shake before using so that the ingredients mix together. The most common emulsions are salad dressings. Creating recipes through this method is called a permanent emulsion because the ingredients come together and do not separate. They help the two liquids get along better. Egg yolks, mustard, and honey are examples of emulsifiers. You can also add another ingredient called an emulsifier before you mix. A temporary emulsion is a blending of ingredients that eventually separate. 
This breaks the water into teeny tiny droplets that get suspended within the oil, this is known as a temporary emulsion. In cooking, you can just mix the liquids together really well through whisking, blending, or strong shaking. Emulsion is a science term for a combination of two liquids that do not usually mix, such as oil and water.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
